Dry needling is often discussed through a muscular lens, yet growing evidence suggests that fascia itself may be a primary driver of pain and movement dysfunction. Understanding fascial anatomy, innervation, and mechanobiology helps explain why targeting fascial tissue—rather than muscle alone—can produce meaningful clinical change.
Clinical Pearl #1: Fascia Is Not Just a Wrapper
Fascia is commonly described as a passive envelope surrounding muscles and organs. In reality, it is a continuous, three-dimensional connective tissue network that penetrates and interweaves within muscles, tendons, ligaments, and neurovascular structures. Rather than separating systems, fascia integrates them.
Capillaries, lymphatic vessels, and nerve endings travel within the fascial matrix, allowing the transmission of biomechanical, chemical, and electrical signals across tissues. This structural continuity helps explain how localized dysfunction can produce regional or even remote symptoms.
Clinical Pearl #2: Fascia Is Highly Innervated—and Often Overlooked
One of the most clinically relevant properties of fascia is its dense sensory innervation. Multiple studies demonstrate that fascia contains six to ten times more sensory nerve endings than muscle, including a high concentration of mechanoreceptors and unmyelinated C-fibers.
These receptors continuously monitor pressure, stretch, vibration, and nociception, contributing to proprioception and pain perception. Importantly, emerging research suggests that muscle fascia may contain more nociceptors than muscle tissue itself, yet it is frequently excluded or underrepresented in pain research models.
This may help explain why injections or interventions directed at fascia have been shown to provoke greater pain responses than those targeting muscle alone—supporting fascia as a significant nociceptive structure rather than an inert tissue layer.
Clinical Pearl #3: Fascia Plays a Key Role in Force Transmission
Beyond sensation, fascia is critical to force transmission and movement efficiency. It contributes to load sharing between muscles, supports joint stability, and facilitates coordinated movement patterns across regions.
Disruption in fascial continuity—whether from trauma, surgery, repetitive loading, or scarring—can alter force transmission and potentially contribute to persistent pain, impaired movement, or compensatory strategies that fail to resolve with muscle-focused interventions alone.
Clinical Pearl #4: Mechanotransduction Explains Why Fascial Needling May Work
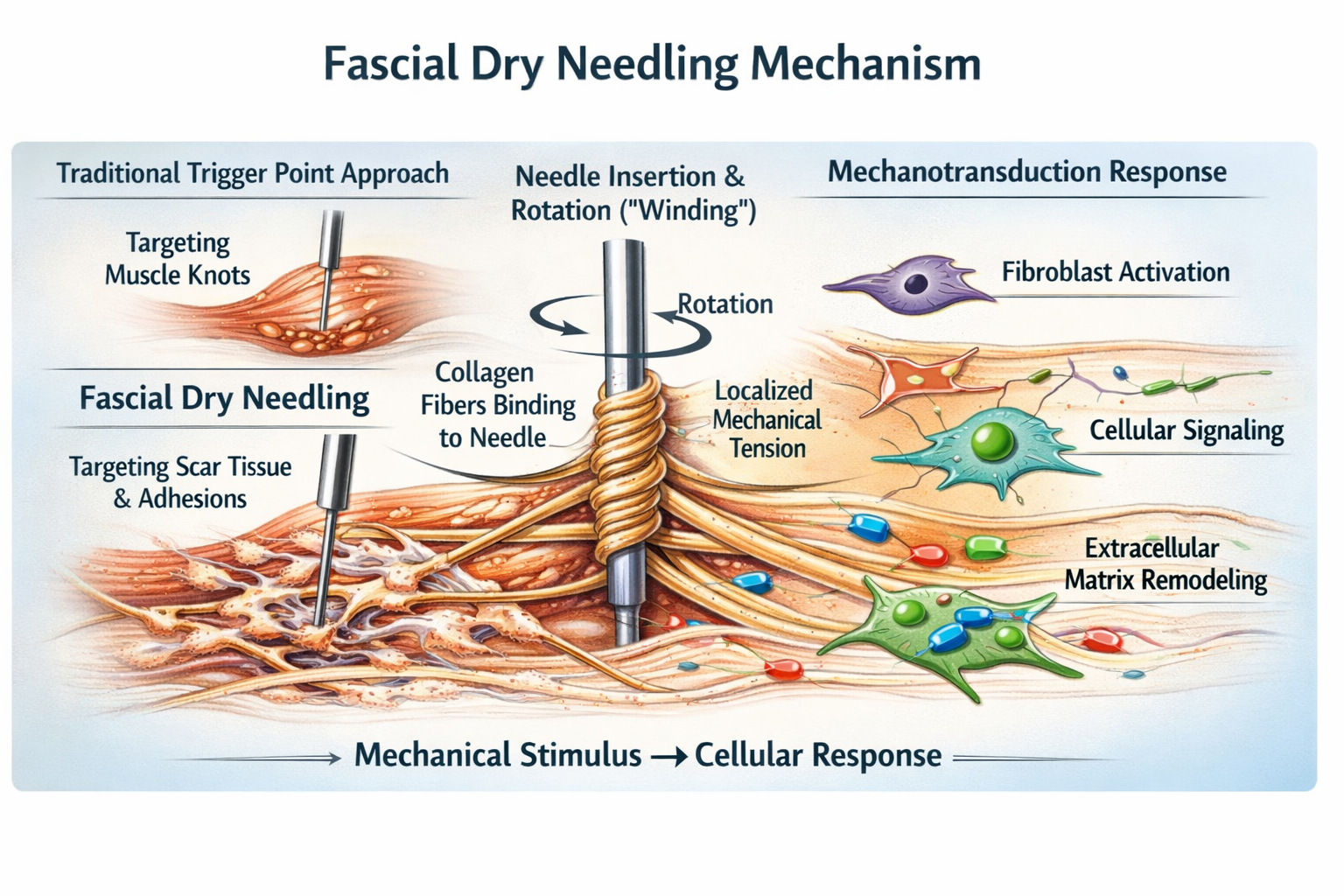
Fascial dry needling differs from traditional trigger point approaches in both intent and tissue response. Using a solid filament needle inserted into fascial tissue, scar tissue, or adhesions, needle rotation (“winding”) creates mechanical coupling between collagen fibers and the needle shaft.
As collagen winds around the needle, localized tissue torque develops. This mechanical stimulus is thought to trigger mechanotransduction—the conversion of mechanical input into cellular signaling—activating fibroblasts and altering the local extracellular matrix.
Clinically, this process may contribute to:
- Analgesic effects
- Reduced tissue stiffness
- Improved tissue glide
- Changes in pain sensitivity
While high-quality clinical trials specific to fascial dry needling remain limited, these mechanobiological principles are well supported in basic science and connective tissue research.
Clinical Applications
In practice, fascial dry needling may be particularly useful for:
- Persistent myofascial pain not responding to muscle-based needling
- Scar tissue and post-surgical adhesions
- Tendinopathies and enthesopathies
- Regional pain patterns with poor tissue extensibility
Case Study #1
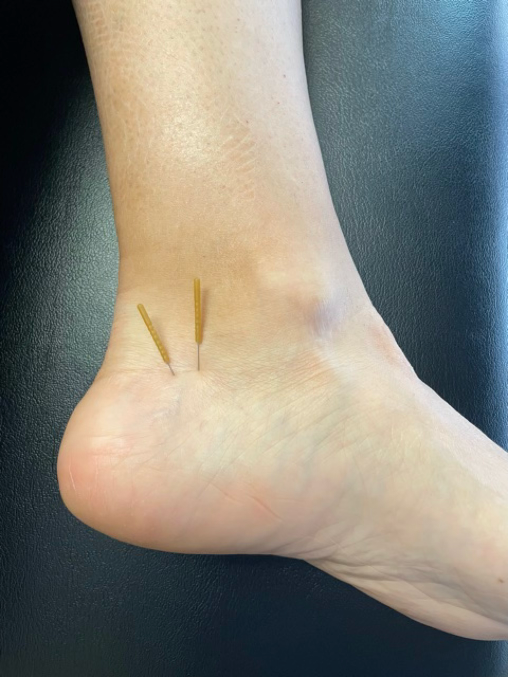
Avid tennis player with an eight-year history of constant medial heel pain unresponsive to years of treatment, including taping, joint mobilization, multiple orthotic interventions, and consultations with foot specialists. Pain-free after 2 fascial treatments.
Case Study #2
One of our students in a DN-2 course had a cesarean section scar (covered by a tattoo) with restricted fascial mobility. During fascial needling of her scar, it reproduced the familiar chronic hip pain she had since her daughter was born eight years ago.
When she returned to attend the DN-3 course a few months later, she reported that her hip pain had not returned since that single intervention.

Case Study #3
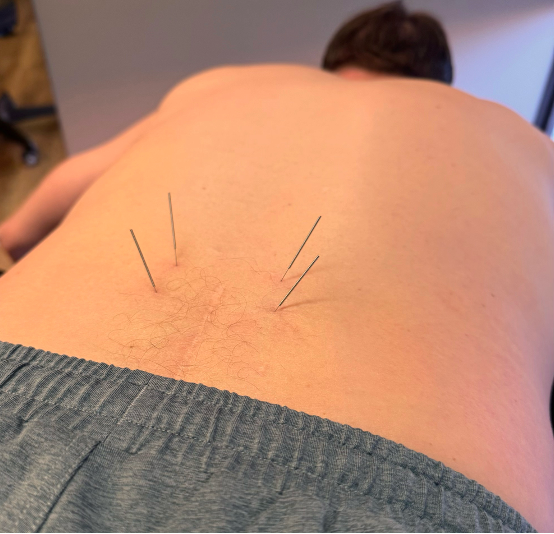
Chronic LBP case – multiple failed surgeries and removal of posterior lumbar instrumentation. His lumbar tissue was so rigid that it could not be penetrated with anything longer than a 30mm needle.
After a series of fascial needling (which reproduced his “familiar pain”), the tissue was pliable enough to use longer 50-60mm needles without a problem, providing significant relief.
Conclusion
Fascial tissue is increasingly recognized as a sensory-rich, force-transmitting, and biologically active system with clear relevance to pain science. While clinical research on fascial dry needling is still emerging, the anatomical and mechanobiological rationale is strong. Incorporating fascial considerations into dry needling practice may help clinicians address pain presentations that persist despite conventional approaches.
Savas Koutsantonis, PT, DPT, CMTPT – Instructor
Select References
Fede C, Clair C, Pirri C, Petrelli L, Zhao X, Sun Y, Macchi V, Stecco C. The Human Superficial Fascia: A Narrative Review. International Journal of Molecular Sciences. 2025; 26(3):1289. https://doi.org/10.3390/ijms26031289
Suarez-Rodriguez V, Fede C, Pirri C, Petrelli L, Loro-Ferrer JF, Rodriguez-Ruiz D, De Caro R, Stecco C. Fascial Innervation: A Systematic Review of the Literature. International Journal of Molecular Sciences. 2022; 23(10):5674. https://doi.org/10.3390/ijms23105674
Gibson W, Arendt-Nielsen L, Taguchi T, Mizumura K, Graven-Nielsen T. Increased pain from muscle fascia following eccentric exercise: animal and human findings. Exp Brain Res. 2009 Apr;194(2):299-308. doi: 10.1007/s00221-008-1699-8.
Findley TW. Fascia Research from a Clinician/Scientist’s Perspective. Int J Ther Massage Bodywork. 2011;4(4):1-6. doi: 10.3822/ijtmb.v4i4.158.
Wilke J, Schleip R, Klingler W, Stecco C. The Lumbodorsal Fascia as a Potential Source of Low Back Pain: A Narrative Review. Biomed Res Int. 2017;2017:5349620. doi: 10.1155/2017/5349620
Schilder A, Hoheisel U, Magerl W, Benrath J, Klein T, Treede RD. Sensory findings after stimulation of the thoracolumbar fascia with hypertonic saline suggest its contribution to low back pain. Pain. 2014 Feb;155(2):222-231. doi: 10.1016/j.pain.2013.09.025.
Adstrum, S., Hedley, G., Schleip, R., Stecco, C. and Yucesoy, C.A. (2017) Defining the Fascial System. Journal of Bodywork and Movement Therapies, 21, 173-177.
https://doi.org/10.1016/j.jbmt.2016.11.003